- Home
- Health Care for Special Conditions
- Cardiopulmonary Rehabilitation
- Medicine in Exercise and Sport
- Health Care in Exercise and Sport
- Guidelines for Pulmonary Rehabilitation Programs

For this revised fifth edition, AACVPR has assembled an expert team of nationally and internationally recognized authors. Together, they present the individuated and interrelated components of pulmonary rehabilitation, including initial and ongoing assessment, collaborative self-management education, exercise training, psychosocial support, and outcome measurement. Highlighted guidelines have been included throughout the book, giving readers easy-to-find guidance for implementing treatment programs and helping patients stay on track.
Guidelines for Pulmonary Rehabilitation Programs, Fifth Edition With Web Resource, offers an evidence-based review in several areas based on the rapid expansion of high-quality scientific evidence since the last edition. To learn best practices for care and program development, readers will find contributions from expert pulmonary rehabilitation professionals in nursing, medicine, physical therapy, respiratory therapy, and exercise physiology. This text provides a scientific, practical framework to help aspiring and current practitioners do the following:
- Understand the current requirements for accrediting pulmonary-based facilities
- Design, implement, or update accredited pulmonary rehabilitation programs
- Deliver optimal care to patients with symptomatic respiratory needs
- Address program issues in exercise, outcomes, and management of pulmonary-based programs
With continued advancements in the science, application, and credentialing of pulmonary rehabilitation programs, the fifth edition of Guidelines for Pulmonary Rehabilitation Programs will assist respiratory practitioners in remaining up to date on the best practices in the field. This edition supports practitioners in understanding the components of pulmonary rehabilitation and applying best practices as well as updating and improving their programs to meet AACVPR certification requirements.
Brian Carlin
Definition
Rationale for Pulmonary Rehabilitation
Pulmonary Rehabilitation and Integrated Care of the Respiratory Patient
History of Pulmonary Rehabilitation
Evidence-Based Guidelines on Pulmonary Rehabilitation
Positioning Pulmonary Rehabilitation within the GOLD 2017 Guidelines
Current Status of Pulmonary Rehabilitation
Summary
Chapter 2. Selecting and Assessing the Pulmonary Rehabilitation Candidate
Gerilynn Connors and James Lamberti
Patient Selection
Patient Assessment
Summary
Chapter 3. Exercise Assessment and Training
Chris Garvey, Rebecca Crouch, and Dave Verrill
Rationale for Exercise Training in Chronic Lung Disease
Mechanisms of Exercise Intolerance in Chronic Respiratory Disease
Exercise Assessment
Field Tests (Walking Tests)
Graded Exercise Test and Cardiopulmonary Exercise Tests
Functional Performance Assessment
Exercise Prescription
Volume, Pattern, and Progression of Exercise Training
Resistance Exercise Testing
Other Tests of Muscular Fitness
Inspiratory Muscle Training
Flexibility Training
Patient Safety
Precautions in Pulmonary Hypertension
Exercise Induced Hypoxemia
Home Exercise Considerations
Emergency Procedures
Documentation of the Evaluation and Treatment Session
Summary
Chapter 4. Collaborative Self-Management and Education
Gerene Bauldoff, Jane Knipper, and Debbie Koehl
Developing an Individualized Self-Management Program
Implementing Self Management Training
Summary
Chapter 5. Psychosocial Assessment and Intervention
Maria Buckley and Kent Eichenauer
Assessment of Psychosocial Functioning
Interventions to Improve Psychosocial Functioning
Partnering with a Psychosocial Provider
Summary
Chapter 6. Nutritional Assessment and Intervention
Ellen Aberegg
Compromised Nutritional Status in Pulmonary Disease
Diet Intake and COPD
Assessment of Nutritional Status
Nutrition Support
Summary
Chapter 7. Patient-Centered Evidence-Based Outcomes
Gerene Bauldoff and Eileen Collins
Timing and Analyzing Outcomes
Patient-Centered Outcome Measures
Summary
Chapter 8. Disease-Specific Approaches to Pulmonary Rehabilitation
Charlotte Tenebeck, Katherine Mensons, Jonathan Raskin, and Brian Carlin
Restrictive Lung Diseases
Pulmonary Hypertension
Other Pulmonary Rehabilitation Populations
Summary
Chapter 9. Program Management and Reimbursement Realities
Trina Limberg, June Schulz, and Karen Lui
Interdisciplinary Team
Program Content and Structure
Administrative Aspects of Program Management
Postrehabilitation Maintenance
Reimbursement Realities
Strategies for Program Success
Summary
Chapter 10. Putting It All Together: Performance Measures, Outcomes, Valid and Reliable Tools, and Program Certification
Anne Gavic and Steve Lichtman
Measuring the Quality of Patient Care
AACVPR Outpatient Pulmonary Rehabilitation Registry
Pulmonary Rehabilitation Certification
Summary
Appendix A: Forms, Questionnaires, Assessments, and Individualized Treatment Plan Example
Appendix B: Excerpt from ATS/ERS Statement: Key Concepts and Advancements in Pulmonary Rehabilitation
Appendix C: Clinical Competency Guidelines for Pulmonary Rehabilitation Professionals
Appendix D: Example of Typical Pulmonary Rehabilitation Facility and Program Schedules
Appendix E: Summary of Additional Recommended Guidelines for Pulmonary Rehabilitation
The American Association of Cardiovascular and Pulmonary Rehabilitation (AACVPR) is dedicated to improving the quality of life for patients and their families by reducing morbidity, mortality, and disability from cardiovascular and pulmonary disease through education, prevention, rehabilitation, research, and disease management. AACVPR is a multidisciplinary professional association composed of health professionals who serve in the field of cardiac and pulmonary rehabilitation. AACVPR also provides educational and professional development opportunities to its members, including cardiovascular and pulmonary physicians, nurses, exercise physiologists, physical therapists, behavioral scientists, respiratory therapists, and nutritionists.
“This book continues to be the bible for evidence-based pulmonary rehabilitation programs.”
—Doody’s Book Review (Four-Star Review)
Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
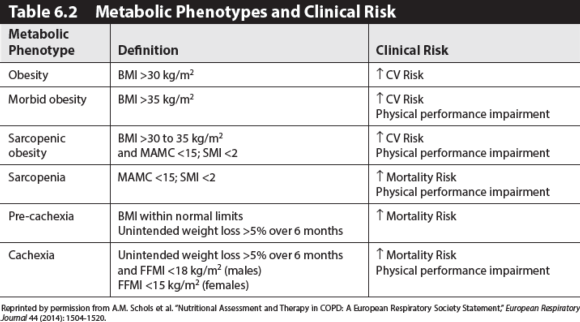
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.
Body Composition Assessment
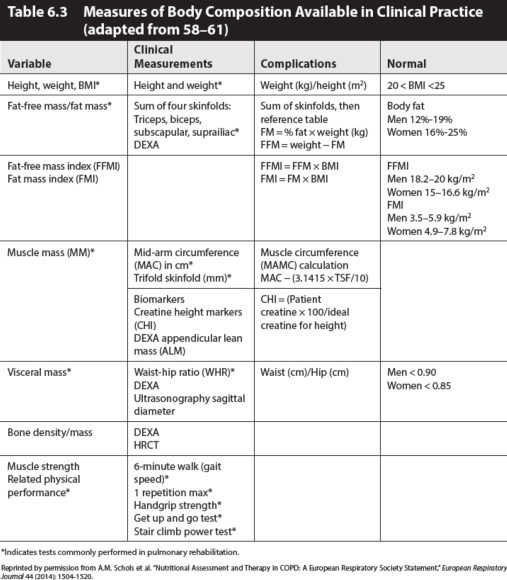
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.
Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.
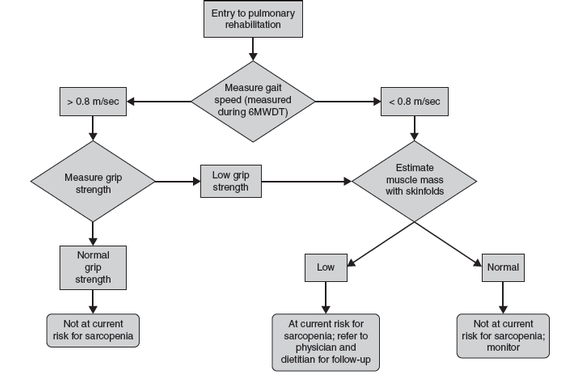
Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
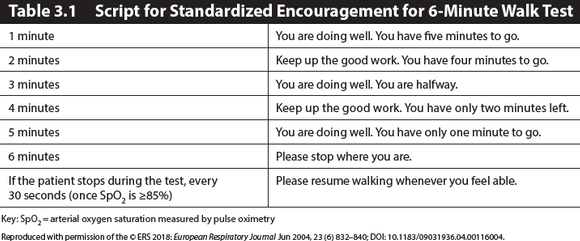
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.
Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
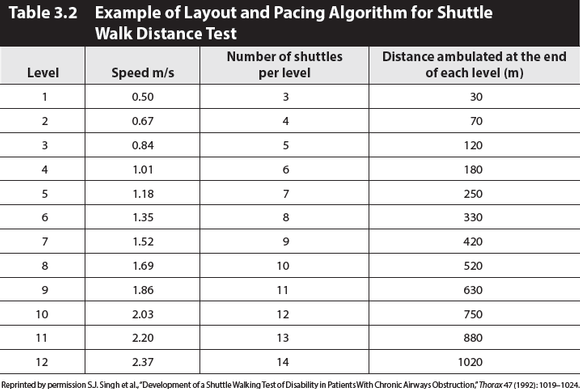
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.
The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui

Assessment of nutritional status in clients with COPD
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake.
By Ellen Aberegg
Assessment of Nutritional Status
Nutritional status is an important determinant of COPD outcome. Evaluation of nutritional status includes analysis of body composition and diet intake. Nutrient status deemed at risk from these two assessments may require further detailed serum analysis. Schols and others have identified different metabolic phenotypes of COPD that are useful in patient counseling as well as in future clinical trial design. Refer to table 6.2 for an overview of the described phenotypes and health risks.

Body Composition Assessment
Incorporation of body composition analysis into nutritional assessment has been a major step forward in understanding systemic COPD pathophysiology and nutritional potential. Identification of patients at risk of diminished fat-free mass increases the potential for improvement in clinical outcomes by subsequent appropriate supplementation. Measurements of height and weight, weight history, and specific anthropometric measurements are assessable in most pulmonary rehabilitation settings. The most accurate methods are not suitable for common daily use in pulmonary rehabilitation due to their high cost and time burden. Skinfolds may overestimate FFM, although many studies found no significant difference by method. Clinical indications of each individual will determine the necessity of more expensive or precise serial methods of assessment, particularly in osteoporotic, pre-cachexic, and cachexic patients. See table 6.3 for assessment information.
Training and competency are essential for pulmonary rehabilitation professionals to ensure accuracy and validity of measures. For specific instructions on methods of measurements, see Fitness Professionals Handbook, 2016. Specific anthropometric measures are listed in table 6.3. Calculations of BMI, FFM, FM, FFMI, and FMI are made using standard reference equations. The major shortcoming of the BMI is that the actual composition of body weight is not taken into account: excess body weight may be made up of adipose tissue or muscle hypertrophy, both of which will be judged as “excess mass.” On the other hand, a deficit of BMI may be due to an FFM deficit (sarcopenia) or a mobilization of adipose tissue or both combined. In a study of European Caucasian males and females aged 18 to 98 years, using the 25th and 75th percentiles as cutoffs, Shutz proposed the following FFMI and FMI ranges as normal. The 25th and 75th percentiles correspond well to the cutoff of BMIs of 20 and 25 kg/m2 respectively. Thus, skinfold measures have been demonstrated as an accessible tool for determination of FFM and FM utilizing the Durnin Womersley equation, the Siri equation, or the Jackson-Pollock equations for men and women. Revised equations for obese individuals have been proposed.
Risk increases as FFM decreases, FM increases, involuntary weight loss occurs, or BMI varies high and low from the norm. As these parameters vary along a continuum, four clusters of risk become apparent. Schols identifies these clusters as metabolic phenotypes: obesity and morbidly obesity, sarcopenia and sarcopenic obesity, normal WNL, plus cachexic and pre-cachexic.

Cachexia and Pre-Cachexia
Cachexia is a complex syndrome frequently present in patients with chronic severe illnesses, including COPD, cancer, and congestive heart failure. Cachexia is associated with increased mortality, impairments in health-related quality of life, and muscle weakness. The clinical phenotype of cachexia ranges from minimal or no weight loss with signs of muscle wasting to severe weight loss with signs of muscle depletion, fatigue, and reduced mobility. The anthropometric measure of FFMI is an indication of risk. An FFMI 2 for men and 2 for women) is a strong predictor of mortality, thus an urgent flag for nutrition intervention. Weight stabilization and gains in FFM will be clear indicators of improvement attainable while the patient is in the pulmonary rehabilitation program. The pre-cachexic patient is in a state of >5% unexplained BW loss over 6 months without measurable deficits in FFM. Nutrition intervention is warranted due to increased risk and weight stabilization and improvement in diet quality will be the indicator of improvement.
Sarcopenia and Sarcopenic Obesity
Sarcopenia is a deficit in skeletal muscle, and thus FFM, resulting in weakness. While the clinical relevance of sarcopenia is widely recognized, there is currently no universally accepted definition of the disorder. Sarcopenia may be optimally defined (for the purposes of clinical trial inclusion criteria, as well as epidemiological studies) using a combination of measures of muscle mass and physical performance.
Sarcopenia is prevalent in patients eligible for pulmonary rehabilitation throughout all ranges of BMI. A deficit of FFM may occur regardless of FM status. Schols recommends using appendicular skeletal muscle index (SMI) (appendicular lean mass as measured by DEXA)/height2)
Sarcopenic obese men had the highest risk of all-cause mortality but not CVD mortality. Efforts to promote healthy aging should focus on preventing obesity and maintaining muscle mass. Abdominal obesity seems to have protective effects on physical functioning. Sarcopenia and central adiposity were associated with greater cardiovascular mortality and all-cause mortality. Atkins found that composite anthropometric measure of MAMC and WC is more effective in predicting all-cause mortality than measures of FFMI and FMI. Traditionally, the nutritional interest in COPD, cystic fibrosis (CF), and lung cancer has been on management of weight loss and muscle wasting (cachexia) in advanced disease stages, while obesity was predominantly investigated in relation to onset and progression of asthma and obstructive sleep apnea syndrome. Obesity has now been identified as a risk factor across all respiratory diseases. Furthermore, combined with aging-induced changes in muscle mass (sarcopenia), the clinical significance of sarcopenic obesity has been highlighted in COPD and lung cancer.
In a large longitudinal study, the Cardiovascular Health Study found that sarcopenic obesity that was characterized using muscle strength was modestly associated with CVD risk. In men with decreased endurance, WC and low muscle mass, as measured using MAMC, were associated with all-cause mortality. Sarcopenia (MAMC ″25.9 cm) and obesity (WC >102 cm) were associated with CVD mortality and all-cause mortality risk, but did not have an excess risk of CVD mortality beyond that associated with sarcopenia or obesity alone. Sarcopenic obesity was more strongly related to non-CVD mortality, independent of inflammation, than CVD mortality. Despite the sarcopenic obese group having the highest risk of mortality, there was no evidence of interaction between sarcopenia and obesity, suggesting that the presence of obesity does not modify the effect of sarcopenia.

Figure 6.1 A suggested algorithm for sarcopenia case finding. Consider comorbidity and individual circumstances that may influence each finding.
Adapted from A.J. Cruz-Jentoft et al., “Sarcopenia: European Consensus on Definition and Diagnosis: Report of the European Working Group on Sarcopenia in Older People,” Age and Ageing 39, no. 4 (2010): 412-423. By permission of A.J. Cruz-Jentoft. Licensed under the terms of the Creative Commons Attribution Non-Commercial License 2.0.
Normal BMI and FFM
Refer to reference ranges for normal BMI, FFM, FM, FFMI, and FMI in table 6.2. Inquiries regarding weight history, including recent changes that were unintended or not desired, should be made in addition to body composition to ensure any “normal” values were not achieved at the expense of FFM loss. Serial measures taken early in the course of pulmonary disease or, at least at the beginning of pulmonary rehabilitation, are helpful in accessing body composition changes over time and as the disease progresses.
Obesity and Morbid Obesity
Obesity, defined as a BMI >25 kg/m2, and morbid obesity, >30 kg/m2, are associated with higher mortality and morbidity due to increase in CVD risk. In nonpulmonary, nonsmoking individuals, a BMI between 20-25 kg/m2 had the lowest all-cause mortality risk. But in COPD patients with moderate to severe obstruction, a BMI 2 was associated with higher risk than those whose BMI >25 or even >30 kg/m2. Although weight loss is routinely recommended for all obese individuals, caution should be exercised in recommending weight loss in COPD patients with moderate to severe obstruction. No studies have systematically investigated the effects of weight loss interventions on adiposity, functionality, and systemic inflammatory profile in patients with COPD. Modest reductions in weight can reduce the cardiovascular disease risk through improvements in body fat distribution. A combination of moderate hypocaloric, moderate to high protein diet and aerobic exercise may achieve this goal best because aerobic exercise training improves insulin sensitivity, induces mitochondrial biogenesis in skeletal muscle, and induces loss of visceral fat mass. Rapid weight loss is frequently associated with loss of FFM and thus is not recommended in pulmonary individuals.
See table 6.4 for metabolic phenotypes and the related nutritional instructions and considerations during pulmonary rehabilitation.
Field (walking) tests for measurement of functional capacity
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting.
By Chris Garvey, Rebecca Crouch, David Verrill
Field Tests (Walking Tests)
Field (walking) tests are commonly used for measurement of functional capacity in the pulmonary rehabilitation setting. Advantages include that the tests are functional, generally safe and easy to perform, inexpensive, and do not require extensive or expensive equipment. These tests are easily administered in the majority of pulmonary rehabilitation patients, including those that are debilitated. However, the test does not offer comprehensive physiological monitoring.
Six-Minute Walk Test
The 6-minute walk test (6MWT) is a widely used outcome measure for pulmonary rehabilitation. The test is safe, easy to administer, involves the use of minimal technical resources, is well tolerated, and accurately reflects a familiar activity of daily living (walking). The 6MWT measures the maximum distance walked in 6 minutes. To obtain valid and reliable results, it is essential to standardize the test procedure, such as staff, configuration of track, patient instructions and verbal reinforcement during testing, type, use and flow rate of supplemental oxygen, and walking aids.
One must consider the energy expenditure changes that occur while performing the 6MWT under certain circumstances, such as while using an assistive walking aid (e.g., rollator) and when a supplemental oxygen source is placed on the walking aid. It has been suggested that there are a reduction of breathlessness and an improvement in the distance walked when using a walking aid or supplemental oxygen. One study found as much as a 20 to 25% energy cost savings with the addition of a walking aid that supported supplemental oxygen. These energy savings appear to be most notable in persons with COPD who have lower exercise performance; e.g., 6MWT distance of
Directions on performing the 6MWT are given below.
Before the Test
- Standardize the shape of the flat walking course: straight versus continuous circle, oval, or square. If using a corridor, a minimum of 30 meters (98.4 feet) that is free of traffic and obstacles should be used.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account (see absolute and relative contraindications, figures 3.4 and 3.5 later in the chapter).
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Rest for at least 10 minutes before beginning the test. If two tests are performed on the same day, at least 30 minutes rest should be allowed between tests.
- Record blood pressure BP, HR, SpO2, and dyspnea score while the patient is sitting or standing. Measure HR and SpO2 continuously during the test.
- Describe the walking track to the patient and give the patient standardized instructions (see table 3.1).
- If the patient has not performed a 6MWT in the recent past, two tests should be performed for initial testing due to a significant learning effect. Use the greater of the two test results.
During the Test
The patient should be asked to walk as far as he or she can in 6 minutes. Standardized encouragement and notification of time elapsed should be given at specific intervals according to the European Respiratory Society (ERS)/American Thoracic Society (ATS) 6MWT script (see table 3.1). Rests are allowed and included in the total test time. If the patient stops during the test (once SpO2 is ≥85%), advise the patient every 30 seconds to “please resume walking whenever you feel able”.
- The total distance covered is recorded.
- The patient is asked to rate shortness of breath and effort using a validated dyspnea and fatigue scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test. If necessary to have staff close to the patient during the walk for safety, they should walk behind the patient.
- Oxygen flow should be held constant during any test; oxygen titration should not be performed during a measured distance outcome walk test. Subsequent tests should use the same flow rate and modality if possible and safe.
- Staff should not assist the patient in carrying or pulling supplemental oxygen unless the patient cannot safely maneuver oxygen.
- Count laps with a lap counter.
- The patient may use their usual ambulatory assistive device; make note of which device (e.g., single-point cane, rollator walker, standard walker) is used and why.
- Monitor the patient for untoward signs and symptoms, including continuous SpO2 and HR.
- The oximeter should not be held by the patient. Either a finger oximeter or a handheld oximeter in a pouch should be used.

Ending the Test
- Once the 6 minutes are completed, the patient is instructed to stop where he or she is.
- Note if the patient exhibits any untoward signs and symptoms.
- Stop the test for:
- SpO2 desaturation <80%
- Chest pain
- Intolerable dyspnea
- Intolerable leg cramps, signs of intolerance to exercise exertion such as staggering, or unusual diaphoresis
- Pale or ashen appearance
- Immediately record SpO2, HR, dyspnea, fatigue, and blood pressure while patient is sitting (measurements taken before and after the test should be done with the patient in the same position).
- Record the distance completed in feet or meters, number of rest stops, total time of stops, and final vital signs including the lowest SpO2 level (and oxygen flow rate if used) on a standardized recording form.
Figure 3.2 shows a sample evaluation form for use in the 6MWT. The MET level corresponding to the average walking speed can be estimated based on the distance walked in 6 minutes. One MET is the amount of energy required while the body is at rest. This may be useful information for assessing exercise capacity and formulating an initial exercise prescription (see figure 3.3). Other information can be gleaned from the test, including gait analysis, balance, fatigue, and pain assessment.
The 6MWT can be influenced by multiple factors, including motivation, encouragement, learning effect, physical course layout, testing environment, gender, and age. Standardized encouragement must be given. There is a learning effect in performing successive 6MWTs, and distances may increase by 26.3 meters on subsequent tests; therefore, two walks should be performed during the initial testing, using the furthest distance of the two tests. If two tests are performed, the recommendation is to wait at least 30 minutes, or up to the next day, to repeat the second test and that the greater of the two values be reported.
The use of a treadmill for 6MWT is not advised. The physical layout of the course may impact walking distance. In the National Emphysema Treatment Trial (NETT), centers that used continuous walking courses, either oval or square, reported longer 6MWT distances than those with straight courses. The difference attributable to the course layout was 110 feet (33.5 meters), or a 10% advantage for continuous courses. The difference between straight and continuous courses may be due to the time and effort required for the subject to turn around on a straight course. The preassessments and postassessments in pulmonary rehabilitation, therefore, must use the same course to eliminate this potential variability.
Prediction formulas for the 6MWT distance are available. This information can be used to assess the patient's level of impairment, compared to data from healthy adults. If using a prediction formula, the formula should be derived populations similar to the pulmonary rehabilitation program population. The minimal clinically meaningful change or minimum clinically important difference (MCID) in the 6MWT distance has been reported to be a mean of 30 meters (98.4 feet). For those patients who walk a very short distance in their initial 6MWT (
Shuttle Walk Tests
There are two types of shuttle walk tests: the incremental and the endurance shuttle walk tests. The ISWT is an incremental, symptom-limited walk test that simulates a symptom-limited cardiopulmonary exercise test (CPET). It measures a symptom-limited walking distance over a marked walking course of 10 meters (33 feet). This distance correlates well with maximal oxygen uptake. The ISWT utilizes an audible pacing timer to incrementally increase the pacing frequency (table 3.2). The subject walks according to the pacing timer frequency until they are too breathless to continue or cannot keep pace with the external pacing signal. Like the 6MWT, the primary test result of the ISWT is the total distance walked. As with the 6MWT, the ISWT should be performed twice prior to an intervention, with the best result recorded. The patient should rest for 30 minutes between tests. Directions for performing the ISWT are given below.

The endurance shuttle test (ESWT) is a standardized, externally controlled, constant-paced walking test for the assessment of endurance capacity. The ISWT is initially performed to determine exercise capacity, and then a paced walk speed corresponding to 85% of walking speed on the ISWT is used to determine the walking speed for the ESWT. The ESWT is a field test equivalent of the constant workload test. The primary outcome of the ISWT is distance, measured to the nearest 10 meters, whereas the primary outcome of the ESWT is time, consistent with other endurance tests.
Since exercise endurance improves more than peak oxygen concentration (peak (O2) after pulmonary rehabilitation exercise training, the ESWT is more responsive to pulmonary rehabilitation than the ISWT. For the ISWT, a change of distance of 47.5 meters has been described as associated with feeling “slightly better,” while a change of 78.7 meters has been associated with feeling “better”; therefore, the MCID for the ISWT distance in COPD is 47.5 meters. Preliminary data suggests the MID for the ESWT is in the region of 180 seconds.
Before the Test
- A medical history for the patient has been reviewed and any precautions or contraindications to exercise testing have been taken into account.
- Instruct the patient to dress comfortably, wear appropriate footwear, and avoid eating or drinking for at least 2 hours before the test (when possible or appropriate).
- Prescribed inhaled bronchodilator medications should be taken if ordered within 1 hour of testing or when the patient arrives for testing.
- Two cones are set 9 meters apart. The distance walked around the cones is 10 meters.
- The patient should rest for at least 15 minutes before beginning the test.
- Record blood pressure, HR, SpO2, and dyspnea score while the patient is sitting or standing.
During the Test
- Standardized instructions should be used via an audio recording.
- No encouragement should be given throughout the test.
- Record each shuttle that is completed on the recording sheet.
- The subject is asked to rate dyspnea and effort using a validated scale.
- The patient should walk alone; staff, other patients, or family should not walk with the patient performing the test.
- Do not assist the patient in carrying or pulling his/her supplemental oxygen.
- The patient may use an ambulatory assistive device; make note of which device (e.g., single-point cane, rollator, standard walker) is used and why.
- A comfortable ambient temperature and humidity should be maintained for all tests.
- Monitor the patient for untoward signs and symptoms.
- Stop the test for:
- Chest pain suspicious of angina.
- Evolving mental confusion or lack of coordination.
- Evolving light-headedness.
- Intolerable dyspnea.
- Leg cramps or extreme leg muscle fatigue.
- Persistent SpO2 ″ 85%.
- Any other clinically warranted reason.
Ending the Test
- The patient is more than 0.5 meter (1.6 feet) away from the cone when the beep sounds (allow one lap to catch up).
- The patient determines that he or she is too breathless to continue.
- For the endurance shuttle walk test (ESWT): The patient reaches 85% of predicted maximum HR.
- The patient exhibits any untoward signs and symptoms.
- Immediately record SpO2, HR, and dyspnea rating while the patient is sitting or standing (measurements taken before and after the test should be done with the patient in the same position).
- Two minutes later, record SpO2 and HR to assess the recovery rate.
- Record the total number of shuttles completed.
- Record the reason for terminating the test.
Adapted from C. Garvey, AACVPR Pulmonary Rehabilitation Outcome Toolkit, https://www.aacvpr.org/Member-Center/Pulmonary-Rehab-Outcomes-Resource-Guide 2017.
Proponents argue that, when compared to a self-paced 6MWT, the shuttle tests are less influenced by motivation or pacing, correlate better with exercise capacity in patients with chronic lung disease, and may be a more sensitive indicator of functional change with rehabilitation or other therapies. Performance on the ISWT relates strongly to direct measures of peak (O2, allowing the prediction of peak (O2. During the ISWT, the cardiorespiratory response to exercise (peak (O2) develops in an incremental fashion similar to formal CPET, thus making this a simple test requiring less technical expertise and equipment. Furthermore, the ISWT is easier to administer, is less expensive, and incorporates an activity that patients perform on a daily basis (i.e., walking).
Strategies to improve success in a pulmonary program
Identify the need for services in the area (competition in the community)
By Trina Limberg, June Schulz, Karen Lui